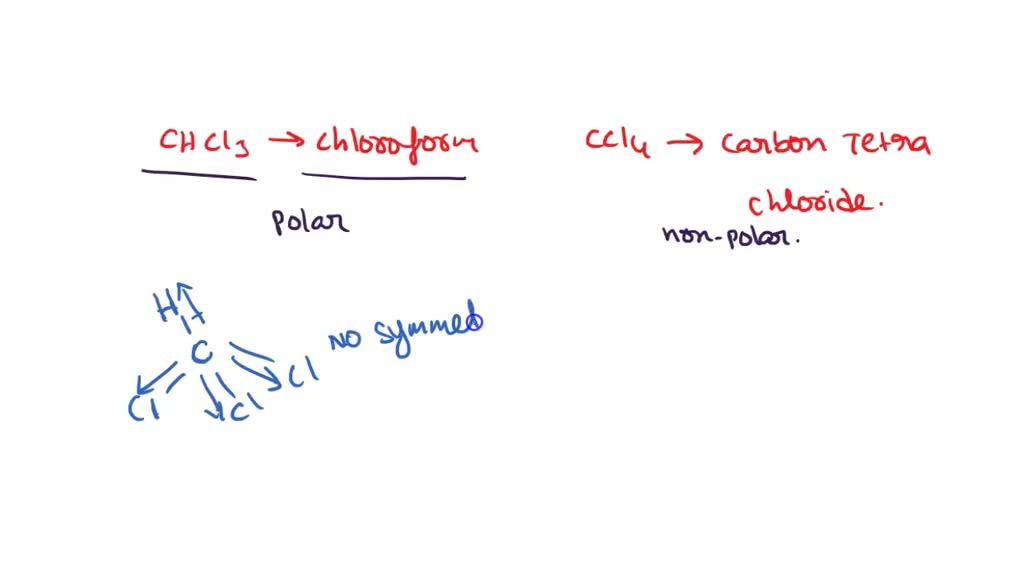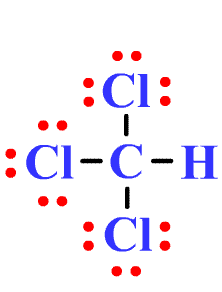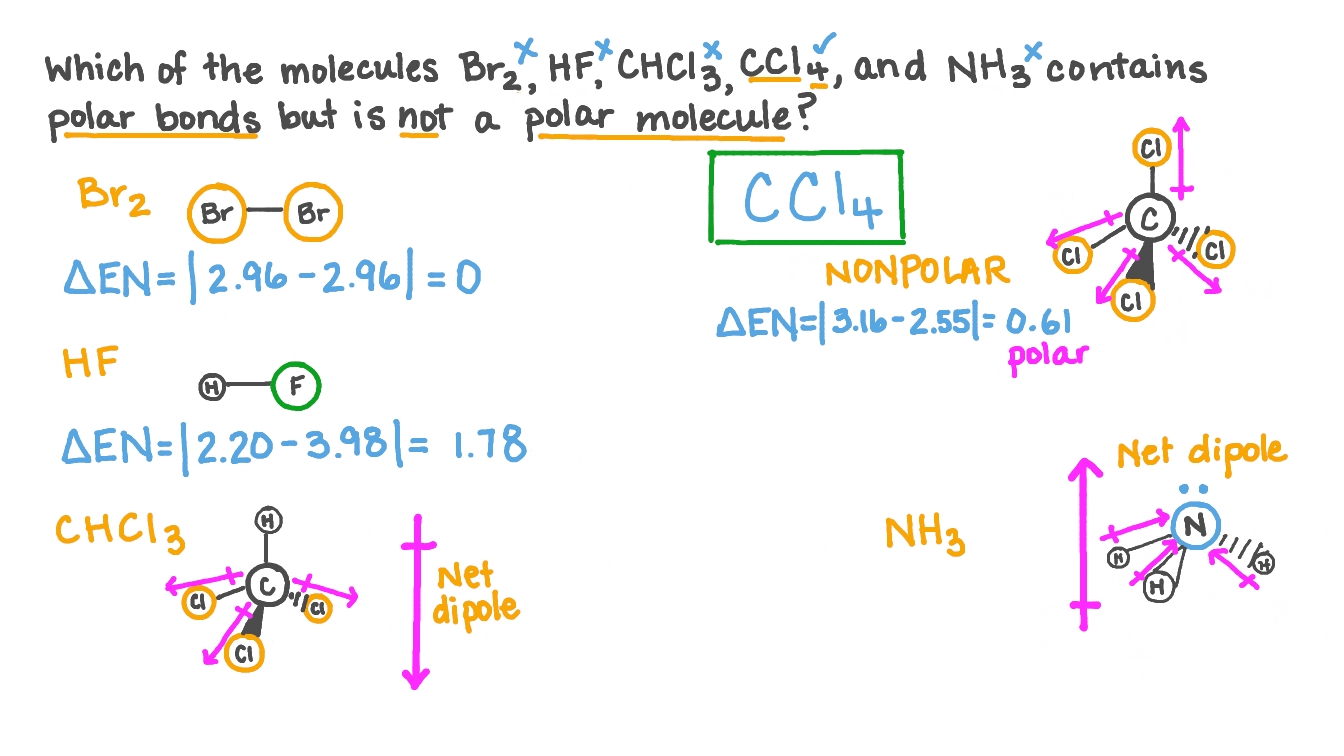
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa
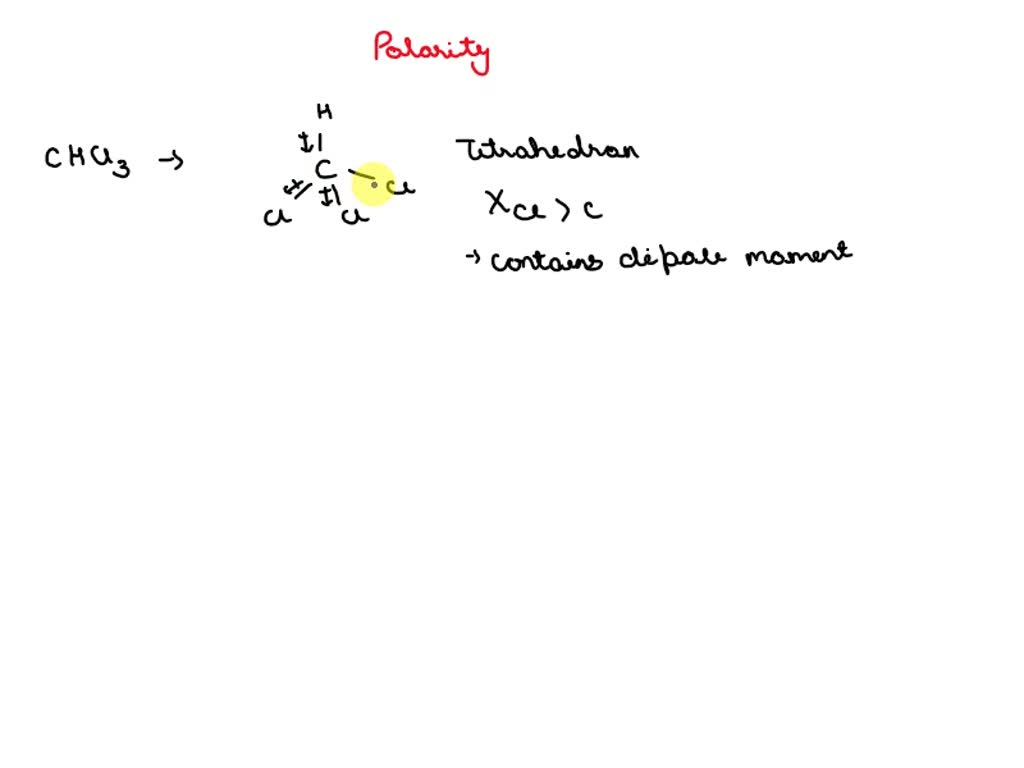
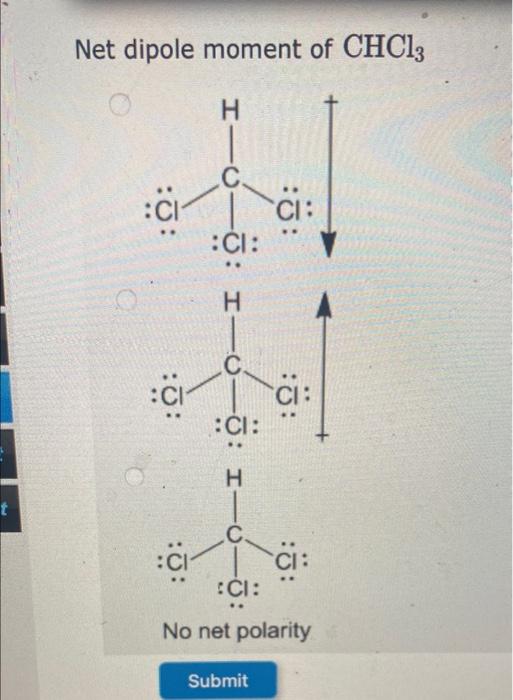
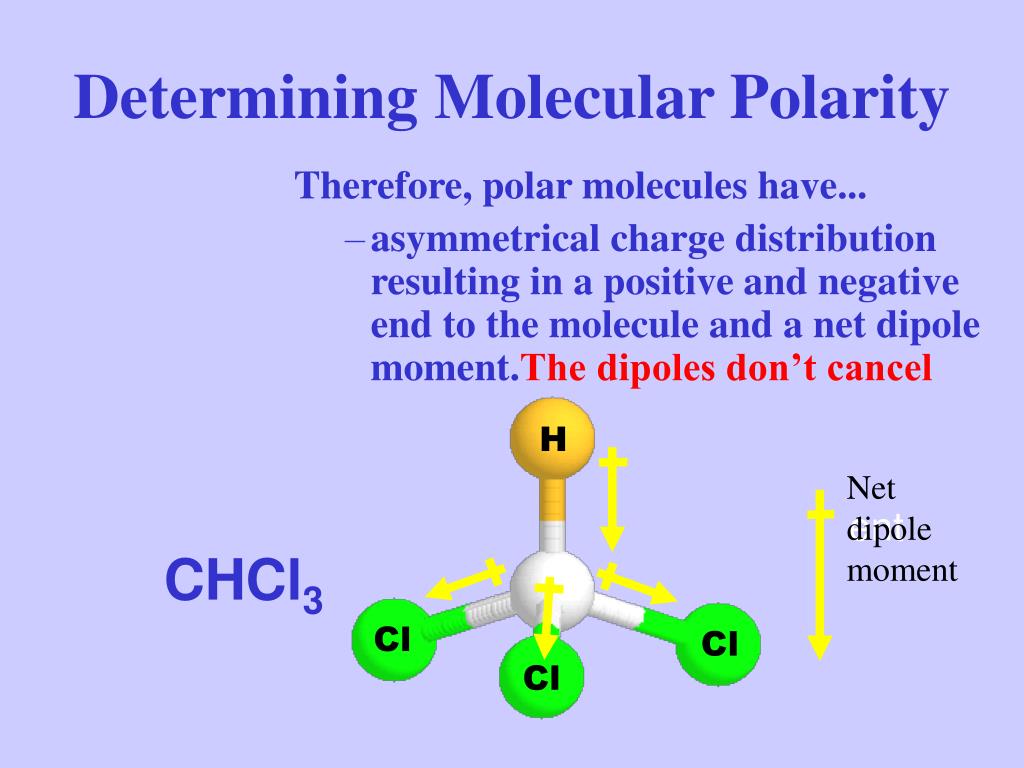
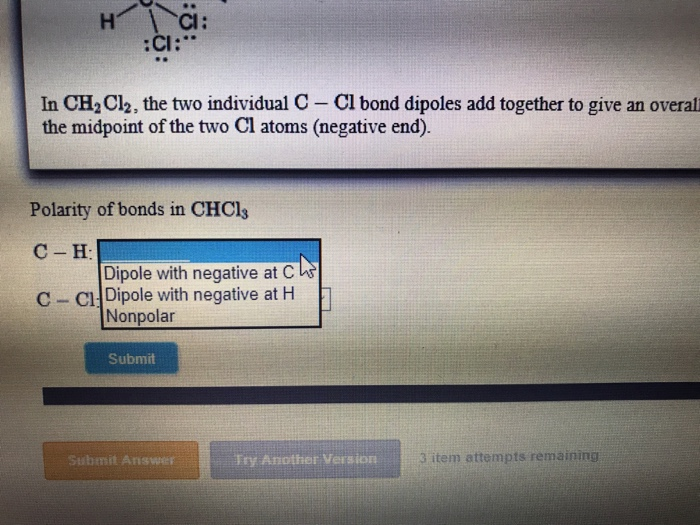
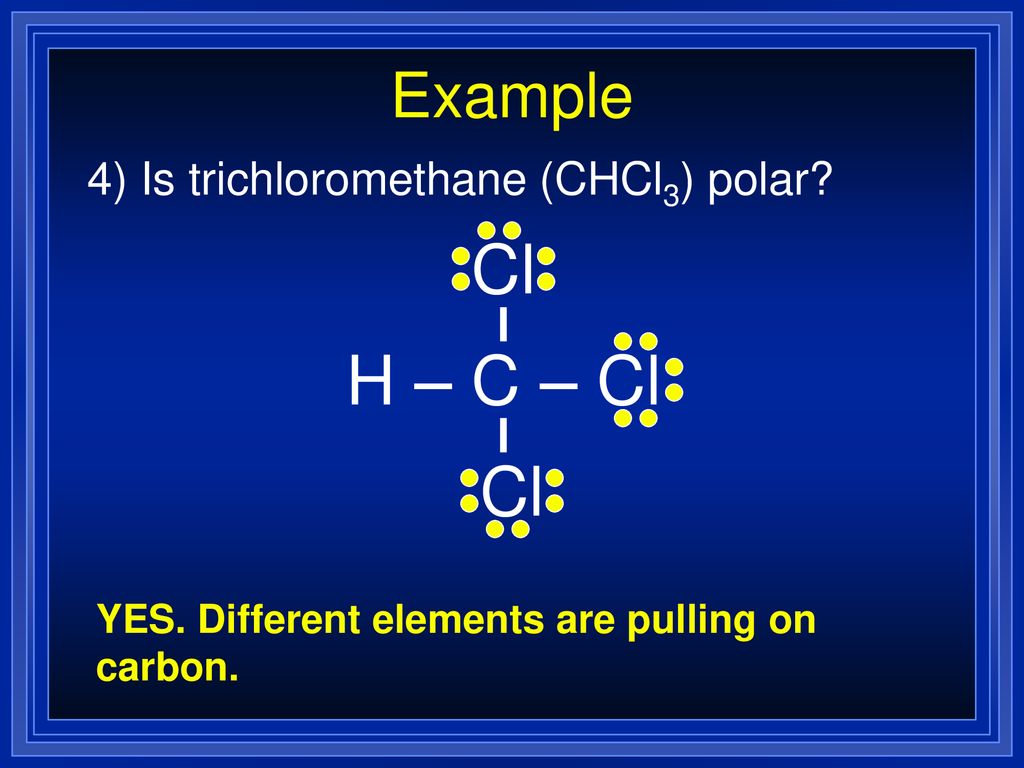
SOLVED: Which statement best describes the polarity of CHCl3 ? a) The molecule is always polar. b) The molecule is always nonpolar. c) Depending on the arrangement of outer atoms, this molecule

Is the molecule CH3Cl polar or nonpolar? If it is polar, specify the direction of its polarity. | Homework.Study.com

Between chloroform and methyl chloride , whose value of dipole moment is greater and why - Chemistry - - 8779339 | Meritnation.com