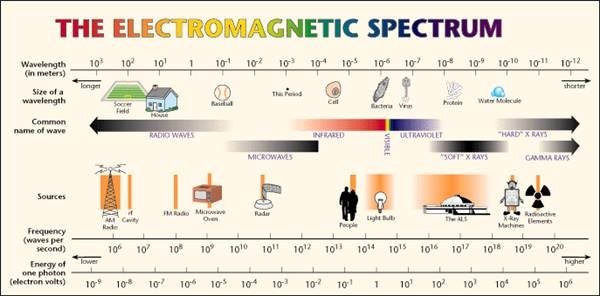
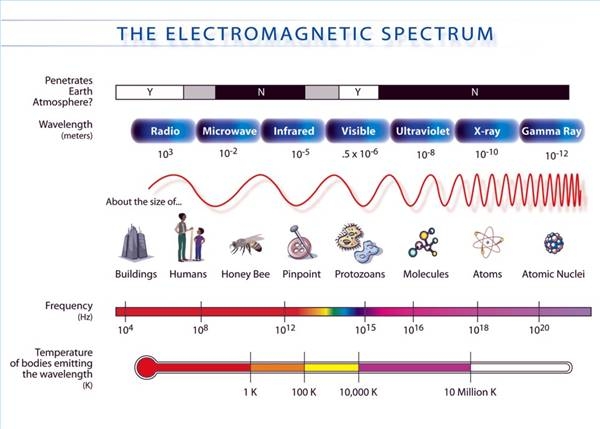
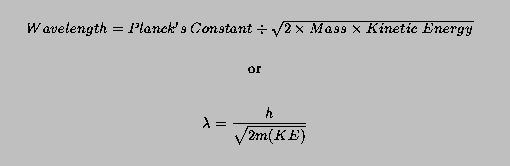Calculate energy of one photon in violet light ,wavelength =360 nanometer Express in eV - Chemistry - Structure of Atom - 2347797 | Meritnation.com

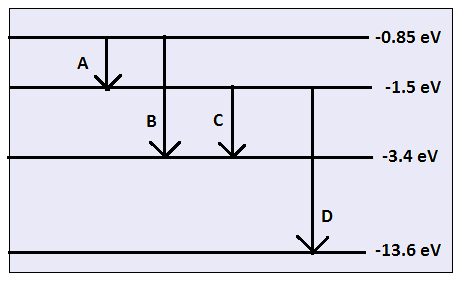
Question Video: Identifying Electron Energy Level Transitions of the Hydrogen Emission Spectrum | Nagwa
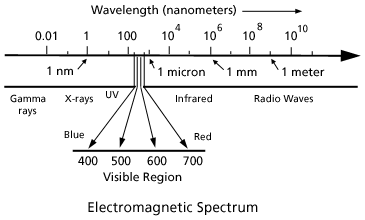
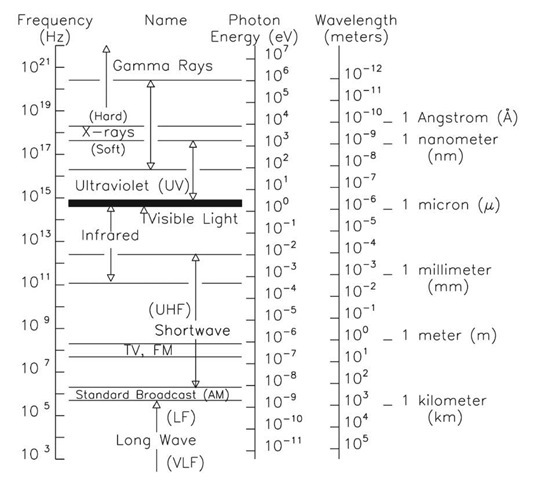
Optical Society of America: Exploring the Science of Light - Teachers and Parents: Articles: Color and Light

The maximum wavelength of light photoelectric effect from a metal is 200 nm. The maximum kinetic energy of electron which is emitted by the radiation of wave length 100 nm will be:

Question Video: Understanding a Photon Emitted By an Electron Transitioning From Energy Level 3 to 2 | Nagwa

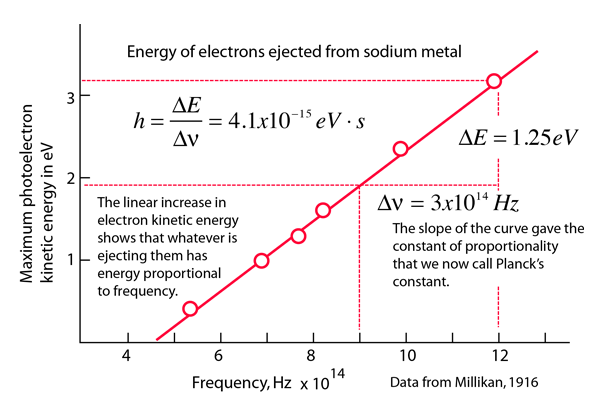
Derivation of hc=1240eVnm from SI units to eVnm + compute the energy of the Lyman-alpha photon in eV - YouTube

Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download

Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download